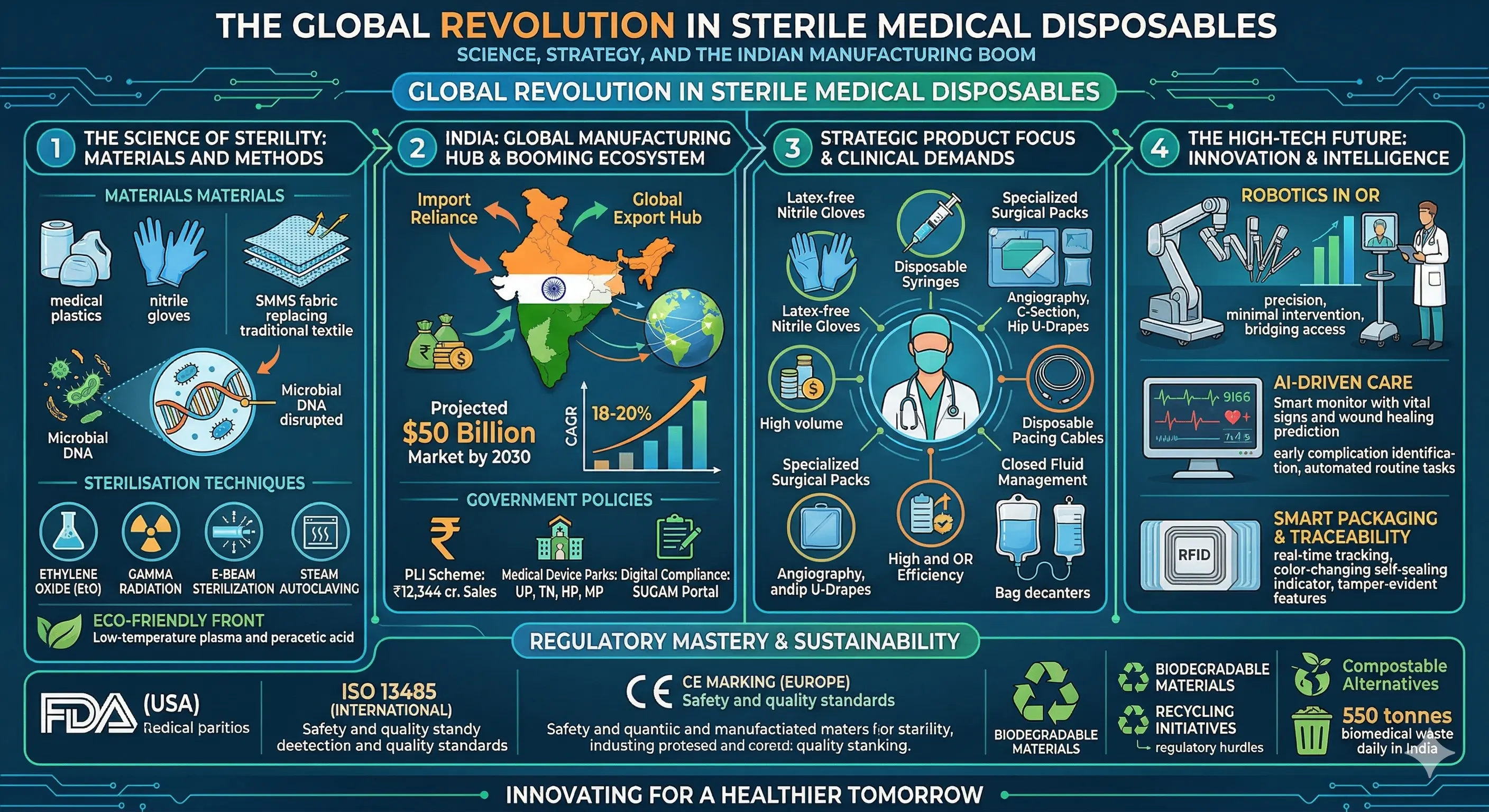
1. The Science of Sterility: Materials and Methods
The effectiveness of any medical disposable begins with Materials Science. Choosing the right material is a delicate balance of biocompatibility, durability, and the ability to withstand rigorous sterilisation without losing structural integrity.
The Core Materials
Manufacturers frequently leverage medical-grade plastics, latex, nitrile, and non-woven fabrics. These materials are chosen for their inert character, which allows them to resist contamination from bacteria and viruses. In India, a significant trend is the shift toward SMMS (Spunbond, Meltblown, Meltblown, Spunbond) fabric, which is replacing traditional textiles in hospitals due to its superior breathability and high-efficiency filtration.
Advanced Sterilisation Techniques
Achieving true sterility requires moving beyond simple cleaning to disrupting the very DNA of microorganisms.
- Ethylene Oxide (EtO) Gas: The gold standard for heat-sensitive devices with plastic or electronic components. It penetrates packaging to disrupt microbial DNA, preventing reproduction.
- Gamma Radiation: Utilises high-energy photons to break DNA strands, making microbes non-viable. This method is effective for both heat-sensitive and non-sensitive bulk items like syringes and surgical gloves.
- Electron Beam (E-Beam) Sterilization: Directs a high-energy electron beam at the product to cause ionisation of microbial DNA. It is prized for its speed and suitability for bulk processing of low-density items.
- Steam Autoclaving: Uses high-temperature steam under pressure to denature microbial proteins. This remains the best choice for heat-resistant surgical instruments.
The Eco-Friendly Frontier: Newer advances like low-temperature plasma reduce energy consumption and thermal damage, while peracetic acid offers a fast process that breaks down into non-toxic substances, addressing the industry's growing environmental footprint.
2. India: From Import Reliance to Global Export Hub
India's medical disposable sector has evolved from a niche market into a booming national ecosystem. The country is rapidly transitioning into a global manufacturing hub, offering cost-competitive alternatives to traditional global supply chains.
The $50 Billion Vision
The Indian medical device market is projected to reach $50 billion by 2030, growing at a staggering CAGR of 18-20%. This growth is structural, driven by the expansion of healthcare into Tier-2 and Tier-3 cities and an increased national focus on infection control.
Policy as a Catalyst
The Indian government has been instrumental in this rise through several key initiatives:
- Production Linked Incentive (PLI) Scheme: This has already led to cumulative sales of over ₹12,344 crore, validating the quality of "Made in India" devices.
- Medical Device Parks: Dedicated clusters in states like Himachal Pradesh, Uttar Pradesh, Madhya Pradesh, and Tamil Nadu are lowering production costs through shared infrastructure.
- Digital Compliance: The CDSCO has streamlined the environment through the SUGAM portal, facilitating risk-based reclassification and automated approvals.
3. Strategic Product Focus: Meeting Clinical Demands
For hospitals and manufacturers alike, focusing on high-demand essentials is key to operational stability.
Critical Consumables
- Gloves and Syringes: These remain the highest-volume items. There is a growing preference for nitrile gloves as hospitals move toward latex-free environments.
- Surgical Drapes and Packs: Specialized packs—such as Angiography, C-Section, and Hip U-Drapes—are essential for streamlining operating room procedures and ensuring a sterile field.
- Pacing and Surgical Cables: Hospitals are increasingly switching to disposable pacing cables (like the ADAP-2000) because they are safer for patients and staff, eliminating the infection risks associated with reprocessing reusable cables.
- Fluid Management: Items like drainage bags and bag decanters are critical for the OR, where closed fluid systems improve efficiency and reduce the risk of spills.
4. The High-Tech Future: Robotics, AI, and Smart Packaging
The next phase of healthcare delivery is being redefined by the integration of intelligence into physical tools.
Robotics in the Operating Room
Robotic systems, such as the Da Vinci®, are setting new world standards by allowing for enhanced precision and minimal intervention. These systems reduce human error, blood loss, and post-surgery complications. Furthermore, telepresence robots are bridging access gaps, allowing specialists to instruct or perform operations in underserved remote areas.
AI-Driven Patient Care
Artificial Intelligence is transforming patient monitoring from a reactive to a predictive process. AI-powered systems can:
- Identify complications early through constant monitoring of vitals and wound healing.
- Automate routine hospital tasks like pharmacy operations and documentation, allowing staff to focus on high-value patient care.
Smart Packaging and Traceability
The sterility of a product is only as good as its packaging. Innovation here includes:
- RFID Tags and Sensors: Enabling real-time tracking of the sterilisation process and inventory management while maintaining sterility to the point of use.
- Self-Sealing Packages: Featuring built-in indicators that change color when the package is fully sterilised.
- Tamper-Evident Features: Distinctive packaging tape makes it immediately obvious if a sterile barrier has been compromised.
5. Regulatory Mastery and Global Standards
Navigating the regulatory landscape is essential for any manufacturer looking to compete globally.
- FDA (USA): Provides the final authority in the U.S., evaluating materials and sterilisation processes before products hit the market.
- ISO 13485: The international standard for quality management systems specifically tailored for medical device manufacturers.
- CE Marking: Necessary for entry into European markets, ensuring products meet stringent safety and environmental requirements.
6. The Sustainability Mandate
With the healthcare industry managing nearly 2 million tons of waste annually, environmental responsibility has become a strategic priority.
- Biodegradable Materials: Research is focused on medical plastics that can break down into water, CO2, and biomass without posing environmental risks.
- Recycling Initiatives: Efforts are underway to strip material from used disposables and return it to service as base units, though regulatory hurdles for recycled medical-grade material remain.
- Indian Waste Management: As India manages 550 tonnes of biomedical waste daily, there is a massive surge in demand for compostable alternatives, such as compostable aprons and garbage bags.
Conclusion: Innovating for a Healthier Tomorrow
The fusion of materials science, advanced sterilisation, and smart technology is doing more than just creating better tools—it is saving lives. As we look toward 2026, the medical disposables industry will continue to be driven by the quest for precision, safety, and sustainability. Whether it is through a perfectly sterilised syringe or a high-precision surgical robot, the goal remains the same: ensuring that every patient receives the highest standard of care, defined by excellence from surgery through recovery.
The Global Revolution in Sterile Medical Disposables: Science, Strategy, and the Indian Manufacturing Boom
The healthcare industry is standing at a crossroads where innovation is no longer a luxury but a life-saving necessity. At the centre of this shift are sterile medical disposables—single-use items ranging from syringes and gloves to advanced pacing cables and surgical drapes—that have become the bedrock of modern infection control. By strictly adhering to Good Manufacturing Practice (GMP) regulations, these products ensure that healthcare-associated infections are significantly reduced, allowing providers to deliver safer care in an increasingly complex clinical environment.
1. The Science of Sterility: Materials and Methods
The effectiveness of any medical disposable begins with Materials Science. Choosing the right material is a delicate balance of biocompatibility, durability, and the ability to withstand rigorous sterilisation without losing structural integrity.
The Core Materials
Manufacturers frequently leverage medical-grade plastics, latex, nitrile, and non-woven fabrics. These materials are chosen for their inert character, which allows them to resist contamination from bacteria and viruses. In India, a significant trend is the shift toward SMMS (Spunbond, Meltblown, Meltblown, Spunbond) fabric, which is replacing traditional textiles in hospitals due to its superior breathability and high-efficiency filtration.
Advanced Sterilisation Techniques
Achieving true sterility requires moving beyond simple cleaning to disrupting the very DNA of microorganisms.
- Ethylene Oxide (EtO) Gas: The gold standard for heat-sensitive devices with plastic or electronic components. It penetrates packaging to disrupt microbial DNA, preventing reproduction.
- Gamma Radiation: Utilises high-energy photons to break DNA strands, making microbes non-viable. This method is effective for both heat-sensitive and non-sensitive bulk items like syringes and surgical gloves.
- Electron Beam (E-Beam) Sterilization: Directs a high-energy electron beam at the product to cause ionisation of microbial DNA. It is prized for its speed and suitability for bulk processing of low-density items.
- Steam Autoclaving: Uses high-temperature steam under pressure to denature microbial proteins. This remains the best choice for heat-resistant surgical instruments.
The Eco-Friendly Frontier: Newer advances like low-temperature plasma reduce energy consumption and thermal damage, while peracetic acid offers a fast process that breaks down into non-toxic substances, addressing the industry's growing environmental footprint.
2. India: From Import Reliance to Global Export Hub
India's medical disposable sector has evolved from a niche market into a booming national ecosystem. The country is rapidly transitioning into a global manufacturing hub, offering cost-competitive alternatives to traditional global supply chains.
The $50 Billion Vision
The Indian medical device market is projected to reach $50 billion by 2030, growing at a staggering CAGR of 18-20%. This growth is structural, driven by the expansion of healthcare into Tier-2 and Tier-3 cities and an increased national focus on infection control.
Policy as a Catalyst
The Indian government has been instrumental in this rise through several key initiatives:
- Production Linked Incentive (PLI) Scheme: This has already led to cumulative sales of over ₹12,344 crore, validating the quality of "Made in India" devices.
- Medical Device Parks: Dedicated clusters in states like Himachal Pradesh, Uttar Pradesh, Madhya Pradesh, and Tamil Nadu are lowering production costs through shared infrastructure.
- Digital Compliance: The CDSCO has streamlined the environment through the SUGAM portal, facilitating risk-based reclassification and automated approvals.
3. Strategic Product Focus: Meeting Clinical Demands
For hospitals and manufacturers alike, focusing on high-demand essentials is key to operational stability.
Critical Consumables
- Gloves and Syringes: These remain the highest-volume items. There is a growing preference for nitrile gloves as hospitals move toward latex-free environments.
- Surgical Drapes and Packs: Specialized packs—such as Angiography, C-Section, and Hip U-Drapes—are essential for streamlining operating room procedures and ensuring a sterile field.
- Pacing and Surgical Cables: Hospitals are increasingly switching to disposable pacing cables (like the ADAP-2000) because they are safer for patients and staff, eliminating the infection risks associated with reprocessing reusable cables.
- Fluid Management: Items like drainage bags and bag decanters are critical for the OR, where closed fluid systems improve efficiency and reduce the risk of spills.
4. The High-Tech Future: Robotics, AI, and Smart Packaging
The next phase of healthcare delivery is being redefined by the integration of intelligence into physical tools.
Robotics in the Operating Room
Robotic systems, such as the Da Vinci®, are setting new world standards by allowing for enhanced precision and minimal intervention. These systems reduce human error, blood loss, and post-surgery complications. Furthermore, telepresence robots are bridging access gaps, allowing specialists to instruct or perform operations in underserved remote areas.
AI-Driven Patient Care
Artificial Intelligence is transforming patient monitoring from a reactive to a predictive process. AI-powered systems can:
- Identify complications early through constant monitoring of vitals and wound healing.
- Automate routine hospital tasks like pharmacy operations and documentation, allowing staff to focus on high-value patient care.
Smart Packaging and Traceability
The sterility of a product is only as good as its packaging. Innovation here includes:
- RFID Tags and Sensors: Enabling real-time tracking of the sterilisation process and inventory management while maintaining sterility to the point of use.
- Self-Sealing Packages: Featuring built-in indicators that change color when the package is fully sterilised.
- Tamper-Evident Features: Distinctive packaging tape makes it immediately obvious if a sterile barrier has been compromised.
5. Regulatory Mastery and Global Standards
Navigating the regulatory landscape is essential for any manufacturer looking to compete globally.
- FDA (USA): Provides the final authority in the U.S., evaluating materials and sterilisation processes before products hit the market.
- ISO 13485: The international standard for quality management systems specifically tailored for medical device manufacturers.
- CE Marking: Necessary for entry into European markets, ensuring products meet stringent safety and environmental requirements.
6. The Sustainability Mandate
With the healthcare industry managing nearly 2 million tons of waste annually, environmental responsibility has become a strategic priority.
- Biodegradable Materials: Research is focused on medical plastics that can break down into water, CO2, and biomass without posing environmental risks.
- Recycling Initiatives: Efforts are underway to strip material from used disposables and return it to service as base units, though regulatory hurdles for recycled medical-grade material remain.
Indian Waste Management: As India manages 550 tonnes of biomedical waste daily, there is a massive surge in demand for compostable alternatives, such as compostable aprons and garbage bags.
.svg)